SEMNI 2022 best thesis award: Dr. Iulen Cabeza
A Numerical Exploration of the Crystalline Lens: from Presbyopia to Cataracts and Intraocular Lenses
Dr. Cabeza-Gil developed various computational methodologies to aid in the treatment of cataracts and enhance our understanding of presbyopia, the two most common eye conditions which are associated with the aging of the crystalline lens.
Presbyopia is a condition that makes it difficult to focus on nearby objects and typically begins around the age of 45, with a prevalence of 100% after the age of 55 1,2. Currently, an estimated 2 billion people worldwide suffer from presbyopia. It is not a medical emergency, but it is a condition that greatly affects people’s quality of life, including reading, computer tasks, etc. On the other hand, cataract surgery is the most frequent surgical procedure performed in developed countries, with 95 million operations annually 3. Cataracts refer to the clouding or loss of transparency of the eye’s natural lens, which is typically replaced during cataract surgery with an artificial intraocular lens.
A better understanding of presbyopia
Dr. Cabeza-Gil developed computational models using the finite element method to better understand the accommodation mechanism (Fig. 1) and conducted ex vivo studies to comprehend the loss of near-focus ability associated with presbyopia. These models, compared with experimental data, allowed the researchers to gain a better understanding of how the crystalline lens changes shape based on its mechanical properties 4,5. For example, the researchers verified that the main cause of presbyopia is the stiffening of the crystalline lens nucleus. They also modeled the active behavior of the ciliary muscle, which is responsible for the accommodative system (Fig. 2).

Fig. 1: Outline of the mechanism of accommodation involving the main elements (lens, zonules and ciliary muscle) in the unaccommodated (left) and accommodated (right) state. The far sight is the unaccommodated state whilst the accommodated state is used to near vision tasks.
The influence of the ciliary muscle on presbyopia is still unresolved. To address this question, Dr. Cabeza-Gil conducted research at the Bascom Palmer Eye Institute (University of Miami, US), where he worked with one of the most powerful groups in the field of presbyopia and obtained more in vivo data about the ciliary muscle. His initial studies suggest that the ciliary muscle continues to function in presbyopic subjects, and that the primary cause of presbyopia is the crystalline lens, as seen in numerical studies. Thanks to the automatic biomedical image segmentation methodologies developed by Dr. Cabeza-Gil in the group 6, large-scale studies could be conducted to determine whether changes resulting from other ocular anatomies, such as the ciliary muscle or sclera, affect presbyopia.

Fig. 2: 3D finite element (FE) model of the accommodative system: lens, zonules, ciliary muscle, iris, sclera and cornea. The arrangement and orientation of the ciliary muscle fibres, longitudinal (yellow), radial (orange) and circumferential (red), is shown on the right.
Towards Personalizing Intraocular Lens Implants in Cataract Surgery
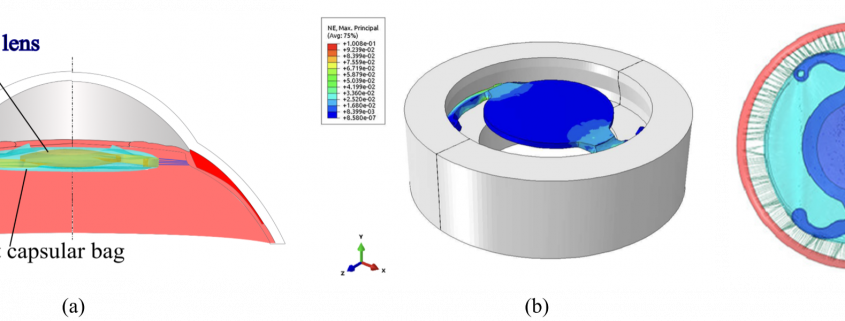
During cataract surgery, an intraocular lens (IOL) is implanted inside the eye to perform the functions of the healthy crystalline lens. The patient’s final visual quality depends on the implanted optical design and the mechanical stability of the IOL inside the capsular bag (Fig. 3a). Small geometric variations can alter optical quality. Dr. Cabeza-Gil developed various computational methodologies experimentally validated to aid in the IOL design process 7. The IOLs must pass various ISO standards tests, including being compressed between two clamps and measuring their biomechanical markers, which are related to the optical performance. Dr. Cabeza-Gil developed a validated finite element model to perform this test (Fig. 3b), which made IOLs safer and enabled manufacturers to check whether their designs behave correctly. Based on this finite element model data, Dr. Cabeza-Gil developed methodologies based on the method design of experiments and deep learning to customize the haptic design based on a patient’s characteristics 8,9. Therefore, given a patient’s characteristics, the IOL with the best mechanical performance could be implanted within the capsular bag.
During his thesis, Iulen also managed to simulate and compare the behavior of the IOL within the capsular bag with experimental data (Fig. 3c) 10. By combining both models and methodologies, they make a very effective and robust tool to be used in improving the efficiency of cataract surgery. These methodologies could, for example, be combined with clinical efficiency-enhancing tools such as SMARTCataract (Alcon, Switzerland) to further improve this surgery. Currently, these implants do not restore the accommodative ability of the eye, but new implants are being developed that require the use of the precise technology developed during Dr. Cabeza-Gil’s thesis to ensure the effectiveness of the surgery.
We hope that you have enjoyed this newsletter on the research conducted by Dr. Iulen Cabeza Gil and his team. Stay tuned for more updates on this fascinating topic!

Fig. 3: (a) Illustrative image of the IOL inside the eye. (b) simulated IOL during the ISO 11979 test. (c) simulated IOL inside the capsular bag.
References
1. Berdahl J, Bala C, Dhariwal M, Lemp-Hull J, Thakker D, Jawla S. Patient and Economic Burden of Presbyopia: A Systematic Literature Review. Clinical Ophthalmology. 2020;Volume 14. doi:10.2147/opth.s269597
2. Fricke TR, Tahhan N, Resnikoff S, et al. Global Prevalence of Presbyopia and Vision Impairment from Uncorrected Presbyopia: Systematic Review, Meta-analysis, and Modelling. Ophthalmology. 2018;125(10). doi:10.1016/j.ophtha.2018.04.013
3. Liu YC, Wilkins M, Kim T, Malyugin B, Mehta JS. Cataracts. The Lancet. 2017;390(10094). doi:10.1016/S0140-6736(17)30544-5
4. Cabeza-Gil I, Grasa J, Calvo B. A validated finite element model to reproduce Helmholtz’s theory of accommodation: a powerful tool to investigate presbyopia. Ophthalmic and Physiological Optics. Published online 2021. doi:10.1111/opo.12876
5. Cabeza-Gil I, Grasa J, Calvo B. A numerical investigation of changes in lens shape during accommodation. Sci Rep. 2021;11(1). doi:10.1038/s41598-021-89145-z
6. Cabeza-Gil I, Ruggeri M, Chang YC, Calvo B, Manns F. Automated segmentation of the ciliary muscle in OCT images using fully convolutional networks. Biomed Opt Express. 2022;13(5). doi:10.1364/boe.455661
7. Cabeza-Gil I, Pérez-Gracia J, Remón L, Calvo B. Effect of haptic geometry in C-loop intraocular lenses on optical quality. J Mech Behav Biomed Mater. 2021;114:104165. doi:10.1016/j.jmbbm.2020.104165
8. Cabeza-Gil I, Ríos-Ruiz I, Calvo B. Customised Selection of the Haptic Design in C-Loop Intraocular Lenses Based on Deep Learning. Ann Biomed Eng. Published online October 2020. doi:10.1007/s10439-020-02636-4
9. Cabeza-Gil I, Ariza-Gracia MÁ, Remón L, Calvo B. Systematic Study on the Biomechanical Stability of C-Loop Intraocular Lenses: Approach to an Optimal Design of the Haptics. Ann Biomed Eng. 2020;48(4). doi:10.1007/s10439-019-02432-9
10. Cabeza-Gil I, Calvo B. Predicting the biomechanical stability of IOLs inside the postcataract capsular bag with a finite element model. Comput Methods Programs Biomed. 2022;221:86-94. doi:10.1016/j.cmpb.2022.106